The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. Practical applications of the series are prediction of double displacement reactions involving two metals and the extraction of metals from their ores. The series is based on empirical data on the ability of a metal to displace hydrogen gas from water and acid. /Lithium-58ee35d95f9b582c4da36490.jpg)
For example, lets say we add calcium to aluminium oxide. The reactivity series is also known as the activity series of metals. Purpose: The purpose of this experiment is to construct an activity series from some metallic elements. A more reactive metal will replace a less reactive one from a compound. After observing or carrying out a series of reactions, you will construct an activity series for some elements. The cookie is used to store the user consent for the cookies in the category "Performance". An activity series allows one to make such predictions. This cookie is set by GDPR Cookie Consent plugin. Build an activity series of metals based upon observations of whether or not a metal reacts with a M2+ aqueous solution. The cookie is used to store the user consent for the cookies in the category "Other. This cookie is set by GDPR Cookie Consent plugin. Activity Series: An easy way to use the table is to note that a metal can reduce any ion of a metal below it in the table. The cookies is used to store the user consent for the cookies in the category "Necessary". The ranking of metals (and hydrogen) based on their activity is called the activity series. The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The activity series is a list of metals and their half-reactions arranged in order of decreasing ease of oxidation or increasing ability to take an electron. The cookie is used to store the user consent for the cookies in the category "Analytics". 
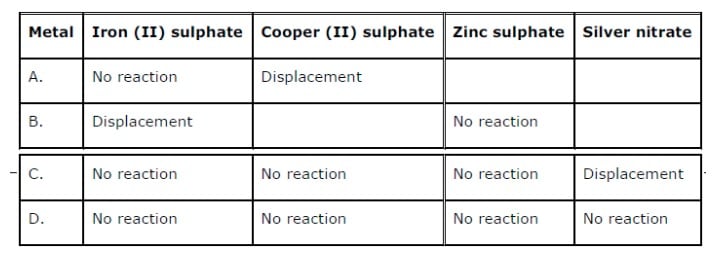
These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. The metals placed above hydrogen in the series can displace it from acids such as HCl and H 2SO 4 (since they are more reactive). Join Seneca to get 250+ free exam board specfic A Level, GCSE, KS3 & KS2 online courses.H + (Non-Metal, Reference for Comparison)ĭespite being a non-metal, hydrogen is often included in the reactivity series since it helps compare the reactivities of the metals. This chemistry video tutorial explains how to determine if a single replacement reaction will proceed as written using the activity series of metals.My Websi. The reactivity of metals is used in order to determine what are the products od single displacement reactions. Want to learn more about Reactivity Series? The activity series of metals relates to the reactivity of metals the order in which they react always from highest to lowest. Because of this, the metal has to be extracted from the ore (rock) where the metal compound is found. Most metals are only found as compounds because the metal has reacted with other elements in the past. Most metals react with dilute acids to produce a salt and hydrogen gas. Potassium, sodium and lithium all react quickly with cold water to produce a metal hydroxide and hydrogen gas. The reactivity series of metals is a list of metals arranged in their order of reactivity from highest to lowest. Platinum, however, is less reactive than copper and so cannot displace copper from a copper sulfate solution. Magnesium + copper sulfate → magnesium sulfate + copper Magnesium is more reactive than copper, so magnesium can displace copper from a copper sulfate solution to create magnesium sulfate. A displacement reaction happens when a more reactive metal (one that forms positive ions more easily) displaces a less reactive metal from a compound. This is called a displacement reaction.Ī metal can only displace another metal from a compound if it is located above it in the reactivity series. The reactivity of metals is due to their incomplete electronic. It is also known by the name activity series. The metals placed above hydrogen in the series can displace it from acids such as HCl and H2SO4 (since they are more reactive). Therefore, the term reactivity series refers to a series of metals arranged in descending order of reactivity. Despite being a non-metal, hydrogen is often included in the reactivity series since it helps compare the reactivities of the metals. These are called native metals.Ī more reactive metal (one that forms positive ions more easily) can displace a less reactive metal from a compound. Reactivity series is an arrangement of metals from highest to lowest reactivity order. Very unreactive metals, such as gold and platinum, are found in the Earth’s crust as pure metals. Metals can be arranged in order of their reactivity. The easier it is for a metal to form its positive ion, the more reactive the metal is. When metals react with other substances, the metal atoms always form positive ions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
